Teacher's Overview
Summary
In this investigation, students are challenged to make careful observations about a burning candle to discover the chemical and physical changes that make it work.
Objective
Students will desribe their observations about a burning candle to discover the chemical and physical changes that make it work.
Safety
- Be sure to wear safety goggles while making close observations, tie your hair back if you have long hair, and always be aware of where the flame is with respect to your clothing—do not lean over the flame.
- Burning candles are open flames that can cause burns. Liquid wax is hot and can cause burns to the skin.
Materials for Each Group
- Tea light candle
- Matches
- Small beaker or evaporating dish (such as 50 mL or 100 mL)
- Balance
- Ruler or meter stick
- String (for measuring circumference)
- Stopwatch
Time Required
One class period, approximately 45–50 minutes.
Lab Tips
Tea candles can be purchased in large quantities, as can tapered candles from hardware stores. Candles of different colors, sizes, and shapes can enrich the observations made by the class.
Student Investigation
We are all familiar with burning candles, but how much have we really seen while looking at one? In this exercise you will be challenged to observe a burning candle through the lens of chemistry. You may be surprised by how much is really happening in what seems like a familiar process!
First we will try to observe details beyond our usual way of looking at a candle, and then we will look for evidence of the physical or chemical nature of any changes that are taking place. Take care in recording your observations—try to imagine that you have never seen a burning candle before!
Obtain a tea light candle, matches, and any measuring equipment available to you. Before lighting the candle, make three qualitative observations and three quantitative observations of the candle.
Qualitative observations describe qualities or characteristics, quantitative observations involve quantities, or measurements. You may choose among many properties to observe: color, texture, mass, physical dimensions, density, aroma… or any others you can think of. Use as many of your physical senses as possible, and be sure to include units for any of your quantitative observations.
Record your obervations
| Qualitative observations | Quantitative observations |
|---|---|
| 1. | |
| 2. | |
| 3. |
Use a match to carefully light the candle.
Repeat the process of making qualitative and quantitative observations of the burning candle, this time making six of each kind of observation. (Some of your observations may relate to properties you have already recorded, that may be changing now that the candle is burning.) Once again use all your senses to make qualitative observations. Ask yourself what you can measure for the quantitative observations.
For your observations and measurements, consider not only the physical candle, but also consider its influence on the air in the space surrounding it.
Record your observations
| Qualitative observations | Quantitative observations |
|---|---|
| 1. | |
| 2. | |
| 3. | |
| 4. | |
| 5. | |
| 6. |
Now sketch a detailed drawing of the burning candle, an enlargement of the portion about a centimeter from the top of the candle to the topmost tip of the flame:
Label the following regions of the candle in your drawing:
- solid wax
- liquid wax
- region of wick without flame
- region of wick with flame
- region of the wick that glows
Now distinguish as many different aspects of the flame itself and label them in the drawing. When you have completed recording your observations and drawings, prepare to blow out the candle, and be ready to make further observations as you do this!
Blow out the candle and record two qualitative and two quantitative observations of the candle immediately after it is extinguished:
| Qualitative observations | Quantitative observations |
|---|---|
| 1. | |
| 2. |
Part One
Now that we have paid close attention to the properties of a burning candle, we can begin to interpret what we’ve seen in terms of phase changes and chemical reactions.
In a phase change (from solid to liquid or from liquid to gas) the particles of a substance (in this case the molecules of wax) will change the arrangement they have with each other. As a solid they will be closely packed together and unable to move freely. In the liquid they will be able to flow around each other but will still be close together, and as a gas they will fly freely apart but they will still be wax molecules and will have the same structure they had as molecules in the solid and the liquid phases:
In the boxes below, use small, single shapes to represent entire wax molecules, and sketch their arrangement in the three physical phases:
Based on your observations of the burning candle, did you find evidence for the change from the solid wax phase to the liquid wax phase?
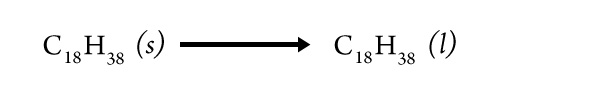
Describe this evidence as specifically as possible.
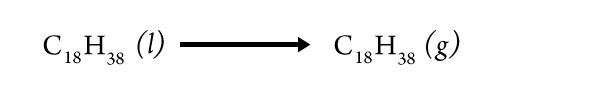
Describe this evidence as specifically as possible.
Did you find evidence for the change from the liquid wax phase to the gaseous wax phase?
- A physical candle is composed of wax surrounding a wick. Look up the definition of the verb “to wick,” and write it down.
- Describe the wicking process in the burning candle, and be sure your description includes the physical state of the wax and the wick.
- As a candle burns, the wax gradually disappears from sight. Is there a physical change in the wax that would account for this disappearance
- If the wax was simply undergoing physical change, would you expect the candle to produce heat and light as it does when a candle burns?
- The process of burning (as opposed to evaporating) is a chemical reaction, a chemical change. The wax molecules are undergoing a chemical change; they are changing into different molecules by reacting with a substance in the air. What is this substance in the air that a candle needs to burn?
Part Two
Interestingly, a candle releases energy through the same kind of reaction that your body uses to obtain energy. Hold your breath for a moment. What is your body craving? This is the same substance the candle needs to burn in a chemical change that we call combustion.
We can represent the combustion of a candle like this:
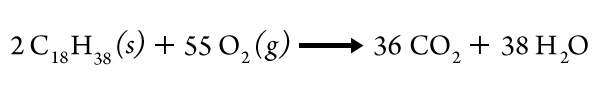
Complete the following sentence that restates this equation:
Two molecules of wax react with ____________ molecules of oxygen to produce__________ molecules of _____________________________ and ___________ molecules of __________________.
We are not aware of oxygen in the air because it is an odorless and colorless gas, and at the temperature of a burning candle, both the CO2 and the H2O are also gases that are invisible to us. We can see evidence of chemical change, however, if we restrict the amount of oxygen that is allowed to react with the wax.
If less oxygen is available, one possible outcome in the combustion of wax is
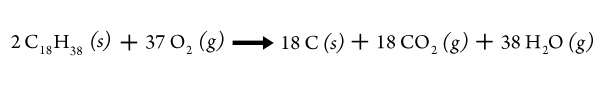
Relight the candle and lower the bottom of a small beaker or a clear watch glass into the flame so that the flame touches the surface of the glass. Hold it there for a moment (where it restricts some of the air flow to the flame) and then remove it.
- What do you observe on the bottom of the beaker? What could this substance be and where did it come from?
- How does this provide evidence that a chemical reaction is taking place?
- Wipe the bottom of the beaker clean and place an ice cube inside it. Hold the beaker several centimeters above (not in) the flame and allow the candle to burn. Can you see a substance forming on the bottom of the beaker? What could this substance be and where did it come from? Could it have come from the melting ice inside the beaker?
- Consider the flame itself. Is the flame a substance produced by the candle? What observations lead you to think that the flame is matter? In what ways does the flame seem like energy?
- Is the following statement true or false: “As a candle burns, wax turns into fire.” If this isn’t an accurate statement, can you restate it correctly?
Part Three
Finally, let us consider the physical phase of the wax as it reacts with the oxygen to produce carbon dioxide and water. Prepare to observe carefully! Blow out the candle and very quickly bring a match close to the wick. Try to notice exactly where the lit wooden splint is with respect to the wick when the candle reignites.
Do this several times to see if you observe something interesting. Note any evidence you have observed that helps to determine the physical phase of the wax when it reacts with oxygen.
- What phase is the wax in when it mixes with oxygen in the air and undergoes a chemical change?
- Based on your observations, in which phase (solid, liquid, or gas) is the wax most likely to undergo a chemical change?
- Explain why this is the case based on your sketches of wax in solid, liquid, and gas phases.
- How might this (your answer to question above) explain a sign that says NO SMOKING near the pumps at a gas station?
- Look back at your diagrams of the candle flame. Can you identify the region where the wick itself is undergoing combustion? Where is this region?
- What is fire? Is it matter or is it energy? Explain your answer.
- In a short paragraph, summarize the changes that a candle undergoes when it burns. Incorporate the following words into the paragraph in a meaningful way: wax, wick, phase change, chemical reaction, vaporize, melt, transforms, solid, liquid, gas, energy, light, and heat.
