Teacher's Overview
Summary
In this investigation, students use canola oil, typically used in the kitchen, to produce biodiesel by reacting it with methanol using a potassium hydroxide catalyst. They then burn a sample of the biodiesel produced. The sample is placed underneath a soda can that contains a measured amount of cold water. The initial and final temperatures of the water are recorded. The collected data is used to calculate the heat of combustion of the biodiesel, which is compared to the theoretical heat of combustion of diesel fuel from crude oil. The concepts of renewable and nonrenewable fuels are also discussed.
Objective
Students learn about a reaction used to produce biodiesel, heat of combustion, and the concepts of renewable and non-renewable fuels.
Materials for Each Group
- 25-mL graduated cylinder
- 10-mL graduated cylinder
- Canola oil (25 mL)
- Methanol (4 mL)
- Plastic container with a screw-top lid (needs a capacity of at least 50 mL)
- Dropper
- 9 M potassium hydroxide (KOH) (12.5 g KOH in 25-mL solution)
- Sodium chloride (0.5 g)
- Large test tube (needs a capacity of at least 30–35 mL)
- Cork or stopper to seal large test tube
- Test tube rack
- 12-oz empty, clean aluminum soft drink can with pull tab
- Ring stand
- Ring
- Thermometer
- Stirring rod
- Matches
- Balance
- Tea light candle with metal cup and wick
- Watch glass
Safety
- Be sure you and the students wear properly fitting goggles.
- Burners must not be used, since many reagents are flammable.
- Methanol particularly poses a serious fire hazard, and its flame is almost invisible. Avoid flames or sparks. Methanol is also toxic by ingestion. Skin contact causes dermatitis. Work in a well-ventilated area.
- When working with alcohols such as methanol, the National Science Teachers Association recommends that “the primary reagent alcohol container be kept in the chemical storeroom. The minimum quantity of alcohol needed for the experiment should be available to students."
- Extreme caution should be used with 9 M KOH. It is caustic and corrosive. The instructor must dispense drops of the concentrated base directly into each group’s container. 9 M potassium hydroxide is caustic and corrosive. Avoid skin contact. In the event of skin or eye contact, rinse well with water. Have contact area evaluated by qualified medical personnel. Instructors should follow their own state’s rules for proper disposal of waste. The waste layer in the investigation contains mostly glycerol, excess methanol, and potentially unreacted potassium hydroxide. A useful document is Biodiesel Safety and Best Management Practices for Small-Scale Noncommercial Use and Production” from Penn State’s College of Agricultural Sciences.
Time Required
Two class periods, approximately 45–50 minutes each. The biodiesel reaction mixture must sit for at least 30 minutes or overnight.
Pre-Lab Discussion
This investigation introduces the concept of heat of combustion of a fuel. It also highlights the difference between renewable and non-renewable fuel sources, in particular, biodiesel and diesel obtained from crude oil. A discussion of the basic organic structures presented in the investigation would be helpful.
Lab Tips
Instead of shaking the bottle with the reaction mixture for 10 minutes, a magnetic stir bar apparatus could be used.
One option for the plastic containers with screw-top lids are “baby soda bottles,” also known as soda bottle preforms. These are sold by science supply companies and look like large plastic test tubes with 2-liter soda bottle screw-top lids.
The reaction mixture is transferred to a test tube after mixing so it is easier to see the two layers separated and to decant the top biodiesel layer. Separatory funnels could be used instead if available. The time needed for the investigation can be shortened if a centrifuge is available; portions of the reaction mixture can be centrifuged to obtain the two separated layers rather than leaving them to separate overnight.
Chilled water should be used in the combustion portion of the investigation. Ice can be added to cool the water if needed. However, unmelted ice should be removed from the water before using it in the soft drink can.
Instructors may wish to use only a few 25-mL graduated cylinders that would be reserved for this activity, as they will become coated with oil and may be difficult to clean thoroughly. Student groups can share the cylinders.
Integrating into the Curriculum
This investigation would fit into units on chemical reactions, thermodynamics, combustion, and green chemistry.
Student Investigation
How did you get to school today? Walk? Ride a bike? Catch a bus? Drive a car? What fuel powered your method of transportation? If you walked or biked, the fuel might have been a bowl of breakfast cereal. Many school buses use diesel fuel, which is made from crude oil that was formed over an incredibly long time by the decomposition of plants and animals. What if instead you could fill your not as crazy as it may at first sound.
These sources of fat or plant oil are potential starting materials for biodiesel, a type of renewable fuel that can be used in diesel engines. Renewable energy sources are those that can be replenished in a short period of time. The materials listed above can be obtained on a much smaller time scale than crude oil; for example, this can be as short as a single growing season for plants.
Crude oil resources, on the other hand, generate over such a long time that they are considered non-renewable.
There are various issues to consider with the different possible materials. For example, certain products that would normally be considered waste, such as used cooking oil headed for the landfill, could be turned into fuel instead. However, even if all the used cooking oil was turned into biodiesel, it would be only a tiny portion of the diesel fuel used in a year. The reaction to form biodiesel from a starting product might create its own difficulties. For example, the reaction that you will use in this investigation produces both biodiesel and glycerol. But, what to do with all the leftover glycerol? It can be used in an application such as soapmaking, but with the large amounts being generated, research is being done for other ways to use it. Canola oil, which comes from the crushed seeds of the canola plant, can be used as a starting material for biodiesel. The structure below shows a typical fat found in canola oil:
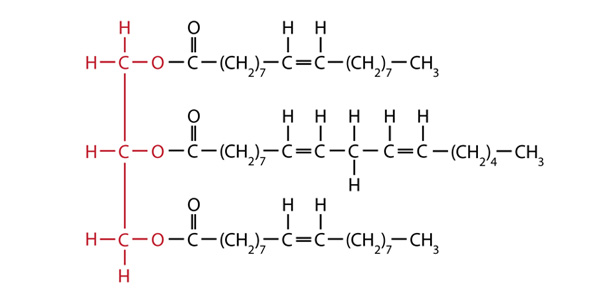
The red portion of the structure represents the glycerol backbone of the fat, while the long portions are the fatty acids that serve as a source of carboxylic acids in the preparation of biodiesel. Biodiesel synthesis breaks the bonds between the glycerol and fatty acids and adds a methyl group (one carbon atom bonded to three hydrogen atoms, —CH3) to the end of the fatty acid.
The other reaction product is glycerol, which can be used in soapmaking. One of the three biodiesel molecules formed from the canola oil molecule above would be:
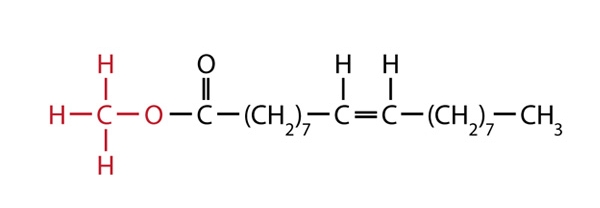
Engines designed to run on diesel fuel made from crude oil can also run on biodiesel fuel with little to no modification, although a blend of diesel and biodiesel is typically used. Diesel fuel is a mixture of hydrocarbons, molecules composed of hydrogen and carbon. Diesel contains molecules with carbon chains of 10–24 carbons. For example, one such molecule might be:
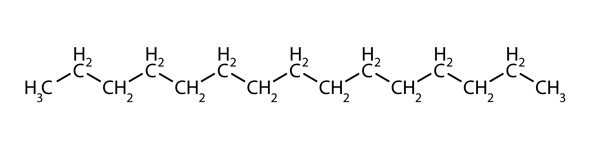
In this investigation, you will use canola oil to prepare biodiesel fuel by combining it with methanol
and a potassium hydroxide catalyst. Then, you will combust a sample of the fuel.
Biodiesel Preparation
- Use a graduated c 1. ylinder to measure 25 mL of canola oil. Pour it into a plastic container with a screw-top lid. Record your observations of the oil’s appearance (color, viscosity, odor).
- Add 4 mL of methanol to the oil in the container. (Caution: Methanol is flammable and toxic. No sources of open flame should be nearby.)
- Ask your instructor to slowly add 5 to 6 drops of 9 M potassium hydroxide to the liquid in the container. (Caution: Potassium hydroxide is corrosive.)
- Tightly cap the plastic container. Shake vigorously for 10 minutes.
- Add 0.5 g sodium chloride to the container. Re-cap and shake vigorously for several seconds.
- Transfer the liquid to a large test tube (should be able to hold at least 30–35 mL). Stopper the top of the test tube with a cork or rubber stopper.
- Allow the test tube to sit in a rack for 30 minutes or overnight. What happens to the contents of the test tube?
Biodiesel Combustion

- Set up the apparatus pictured in the top photograph to measure the heat of combustion of the
biodiesel you prepared.
- Slightly bend up the pull tab of an empty, clean aluminum soft drink can.
- Slide a glass stirring rod through the top hole of the pull tab.
- Hold the glass stirring rod horizontally and set it on a ring attached to a ring stand so the soft drink can is suspended underneath it.
- Raise or lower the ring stand so the bottom of the can is ~2 cm above the wick of the metal sample cup you will use to burn the biodiesel.

- Prepare the sample of biodiesel for combustion as pictured in the photograph at right.
- Take a tea light candle in its metal cup. Remove the candle from the cup.
- Remove the metal circle and its attached wick from the bottom of the candle.
- Set the candle aside. Place the metal circle and its attached wick back in the metal cup, so the wick stands upright.
- Decant ~5 mL of the biodiesel layer (top layer) from the test tube or remove ~5 mL of the top layer using a dropper. Place this sample in the metal cup.
- Dispose of the remaining layer according to your instructor’s directions.
- Record your observations of the sample’s appearance (color, viscosity, odor). Measure and record the initial weight of the biodiesel sample with the cup and wick to the nearest 0.1 g.
- Weigh ~100 g of cold water, recording the weight to the nearest gram. Pour the water into the soft drink can. Measure and record the initial temperature of the water to the nearest degree Celsius.
- Ignite the biodiesel sample using the wick. Once it is ignited, immediately move the metal cup underneath the soft drink can.
- As the water in the can heats, stir it gently. Allow the biodiesel sample to burn for ~5 min.
- Extinguish the flame by placing a watch glass over the metal cup.
- Measure and record the highest temperature reached by the heated water to the nearest degree Celsius.
- Allow the metal cup and sample to cool. Measure and record the final weight of the biodiesel sample with the cup and wick to the nearest 0.1 g.
- Using the temperature and weight data from heating the water in the can, calculate how much thermal energy was used to heat the water. The specific heat capacity of water is 4.18 J/(g · °C), meaning it takes 4.18 J to raise the temperature of 1 g of water by 1°C.
- Calculate the heat of combustion in kJ/g for the sample of biodiesel you burned. The heat of combustion is the quantity of thermal energy given off when a certain amount of a substance burns. Assume that all of the energy released by the burning biodiesel is absorbed by the water.
- Petroleum diesel (from crude oil) produces 43 kJ/g of thermal energy when burned. Compare this to the thermal energy your biodiesel sample produced when it was burned.
- Compare your calculated heat of combustion with those calculated by the rest of the class. What is the class mean?
- What evidence was there that a reaction occurred during the biodiesel preparation?
- Look again at the structures of two possible biodiesel and diesel molecules in the Preparing to Investigate section. What similarities do the molecules have that would help to explain how biodiesel can be used with little to no modification in diesel engines?
- Draw the balanced equation for the reaction to form biodiesel from canola oil.
Practice Calculations
You can get more practice with calculations like these in The Energy Efficiency of Heating Water.
- Is biodiesel a “better” fuel than diesel fuel from crude oil? Explain and support your answer.
- The combustion of biodiesel produces carbon dioxide (CO2), a greenhouse gas. However, biodiesel is labeled as “carbon-neutral,” meaning it does not increase the net amount of carbon (carbon dioxide) in the atmosphere. Why?
- What is a potential concern with diverting food crops to the production of biodiesel?
- Suppose that you wanted to make biodiesel for your own car using the method in this investigation. What challenges might you encounter in scaling up the process?
